The scientific basis and data requirements for dossiers at different stages of development will be communicated for the quality, preclinical and clinical parts of regulatory submissions. Case-studies will be inserted for the practical application of knowledge gained.
The scientific basis and data requirements for dossiers at different stages of development will be communicated for the quality, preclinical and clinical parts of regulatory submissions. Case-studies will be inserted for the practical application of knowledge gained.
The training will be delivered as presentations on individual topics, interspersed by case studies and conclude with an outlook on further developments in this fast moving field.
This 3-day training course focuses on drug development of biopharmaceuticals which have become the focus of pharmaceutical innovation and currently represent 30% to 50% of newly authorised medicinal products.
Participants will learn about the legislative and regulatory framework for biopharmaceuticals in Europe and the roles of the European Medicines Agency and National Competent Authorities in market access. This covers the clinical trial stage, licensing and the life-cycle of these products. Specific development paths will be delineated, such as those for biosimilars and ATMPs, the latter comprising somatic cell therapies, gene therapies and tissue engineered products.
The Development of Biopharmaceuticals
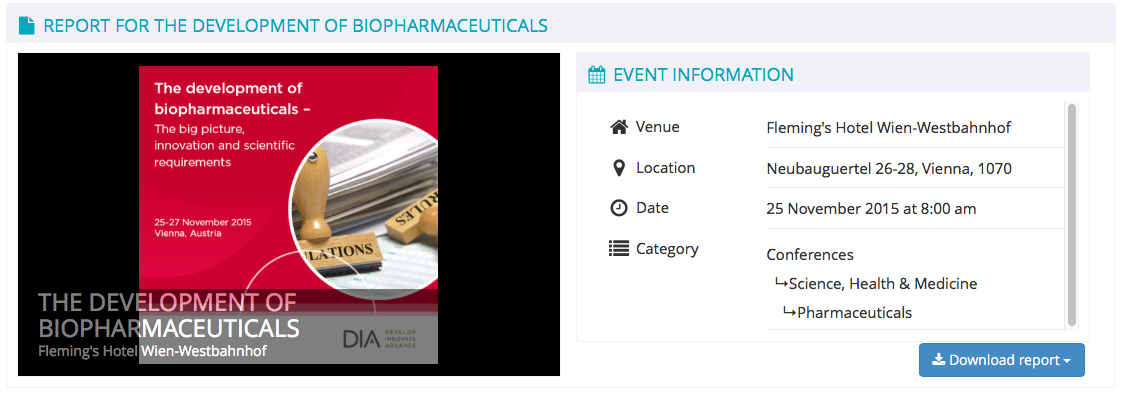
on Wednesday November 25, 2015 at 8:00 am (ends Friday November 27, 2015 at 5:15 pm)
Information :
The development of biopharmaceuticals – The big picture, innovation and scientific requirements
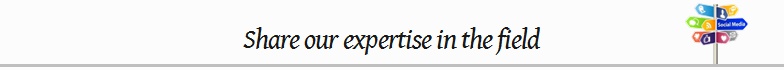
We ensured was distributed across all relative event listing sites locally, nationally, globally and socially with a total site reach of 40.3 M !
Industry: 23
Targeted : 29
Global : 12
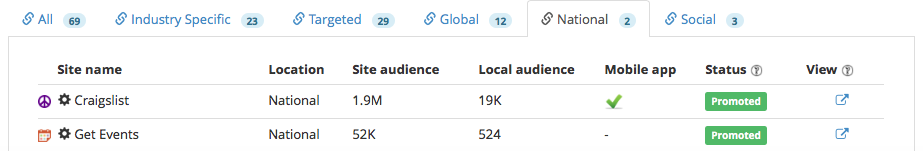
National : 2
Social: 3
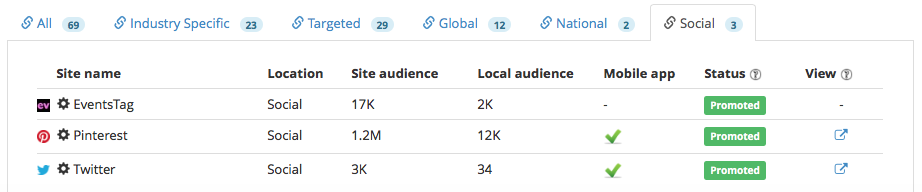
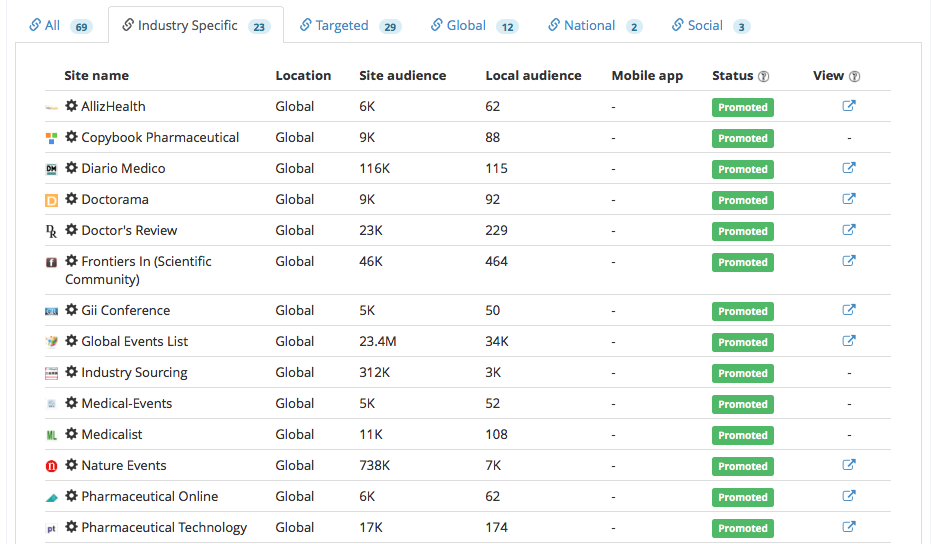
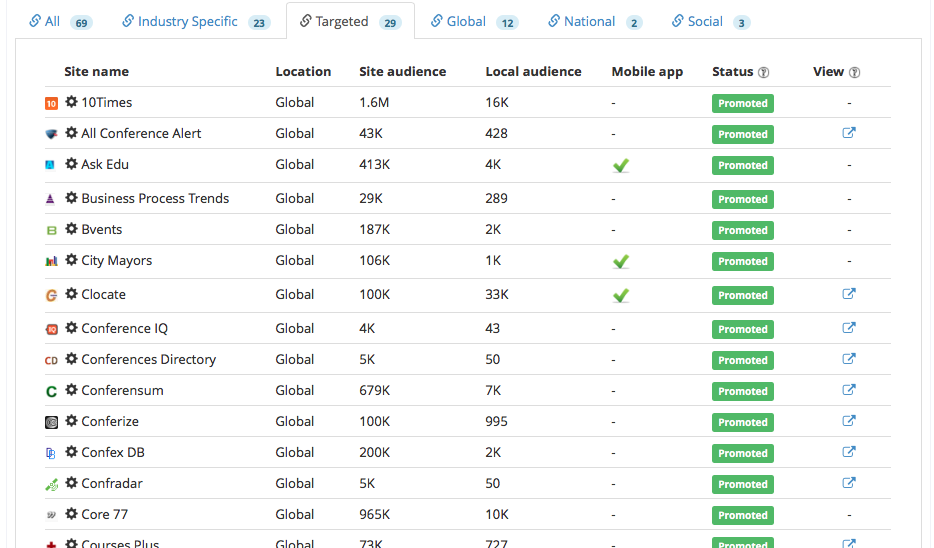
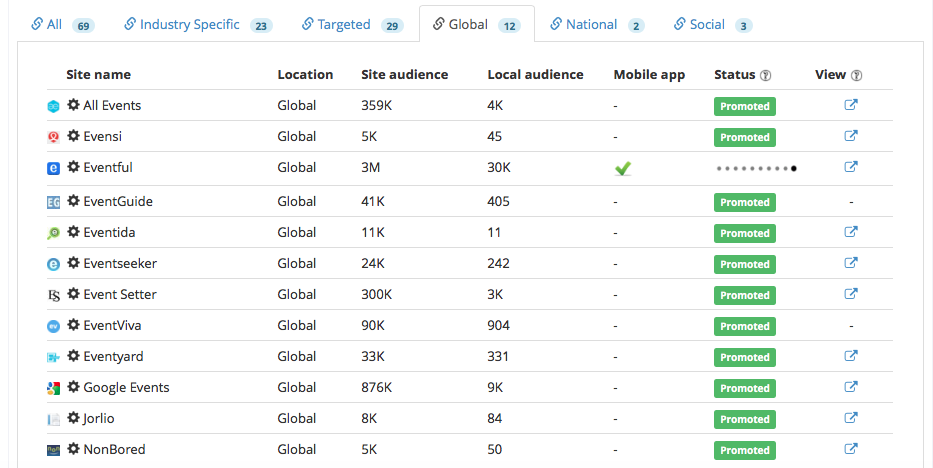
We wanted to reach the maximum number of potential event attendees to participate in the event, we promoted the event to a wide network of industry publishers composed of 23 industry specific, 29 targeted, 12 global, 2 national and 3 social.
The following images are examples of the event listed on targeted listing sites:
![]()
Click on the logos below to view listing results:
We can grow your audience reach and achieve better natural search. The publishers who we work with can expect to appear on the first page of Google. evvnt customers often ask about the impact of the content distribution we provide on their search rankings, and it is important for us to ensure we continue to make a positive impact on their behalf. To learn more click here.
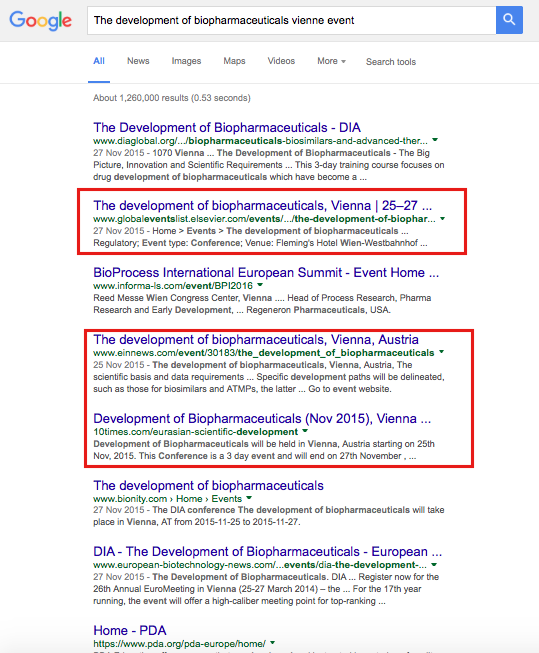
40% first page of Google for the event
To be as transparent as possible, evvnt always sends a detailed report to each client.
Broadcast results: Total number sites the event has been submitted to, depending on the category, location and the level of targeting required for the event.
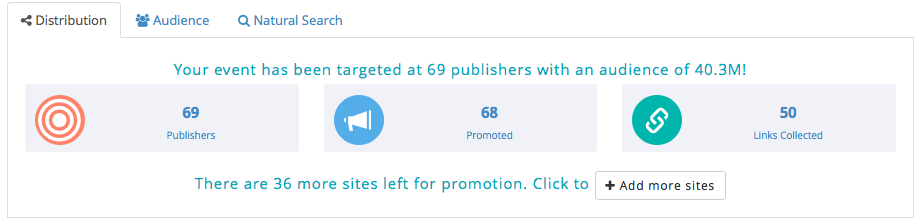
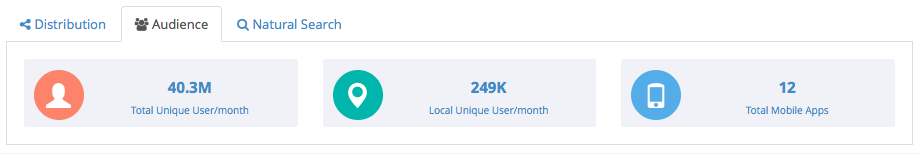
Audience reach: See how many people can see the event including aggregated total unique users globally and estimated to the event. We also document sites that pass content on to mobile apps.
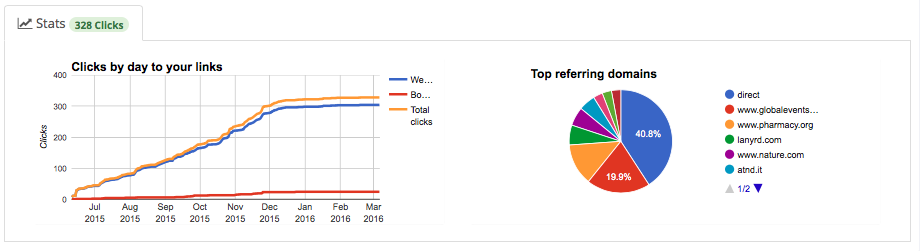
Detailed reporting: A click tracking report with a summary of the events performance is activated and supplied within the product segmented by url type and the site delivering traffic.
Key figures:
72% of live links within 24 Hours
12 mobile apps
Active in all 5 major search engines.
Tweets by @mayocliniclabs
Tweets de @DrugInfoAssn//